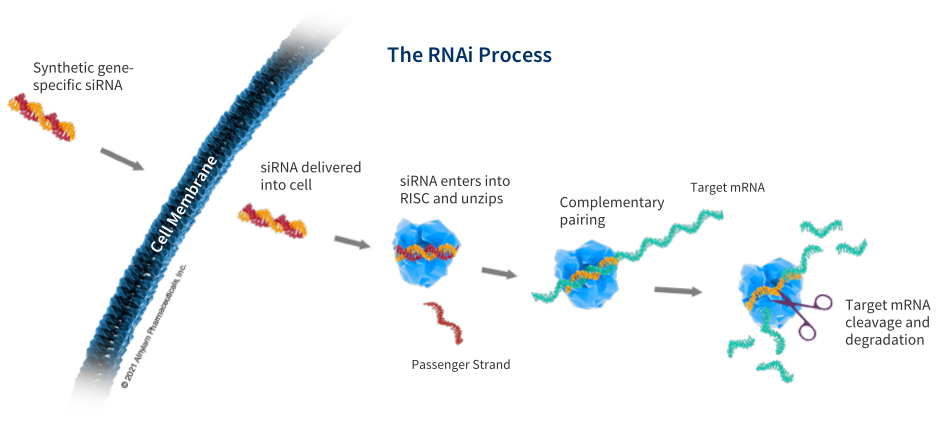
Alnylam Pharmaceuticals on Twitter: "Symptoms of acute hepatic #porphyrias are similar to those of other diseases, resulting in frequent misdiagnosis. Genetic testing through Alnylam Act may help: https://t.co/q9990gYwya https://t.co/oLYz6BU69U" / Twitter

ALNYLAM ACT Trademark of Alnylam Pharmaceuticals, Inc. - Registration Number 5498799 - Serial Number 87340411 :: Justia Trademarks
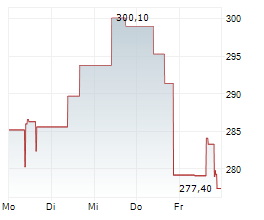
Alnylam Pharmaceuticals, Inc.: Alnylam und Regeneron melden positive klinische Zwischenergebnisse der Phase 1 zu ALN-APP, einem RNAi-Prüftherapeutikum für Alzheimer-Krankheit und zerebrale Amyloidangiopathie

IRA Effect: Alnylam Acting 'Rationally' In Halting Second Orphan Indication For Amvuttra – Analysts :: Pink Sheet