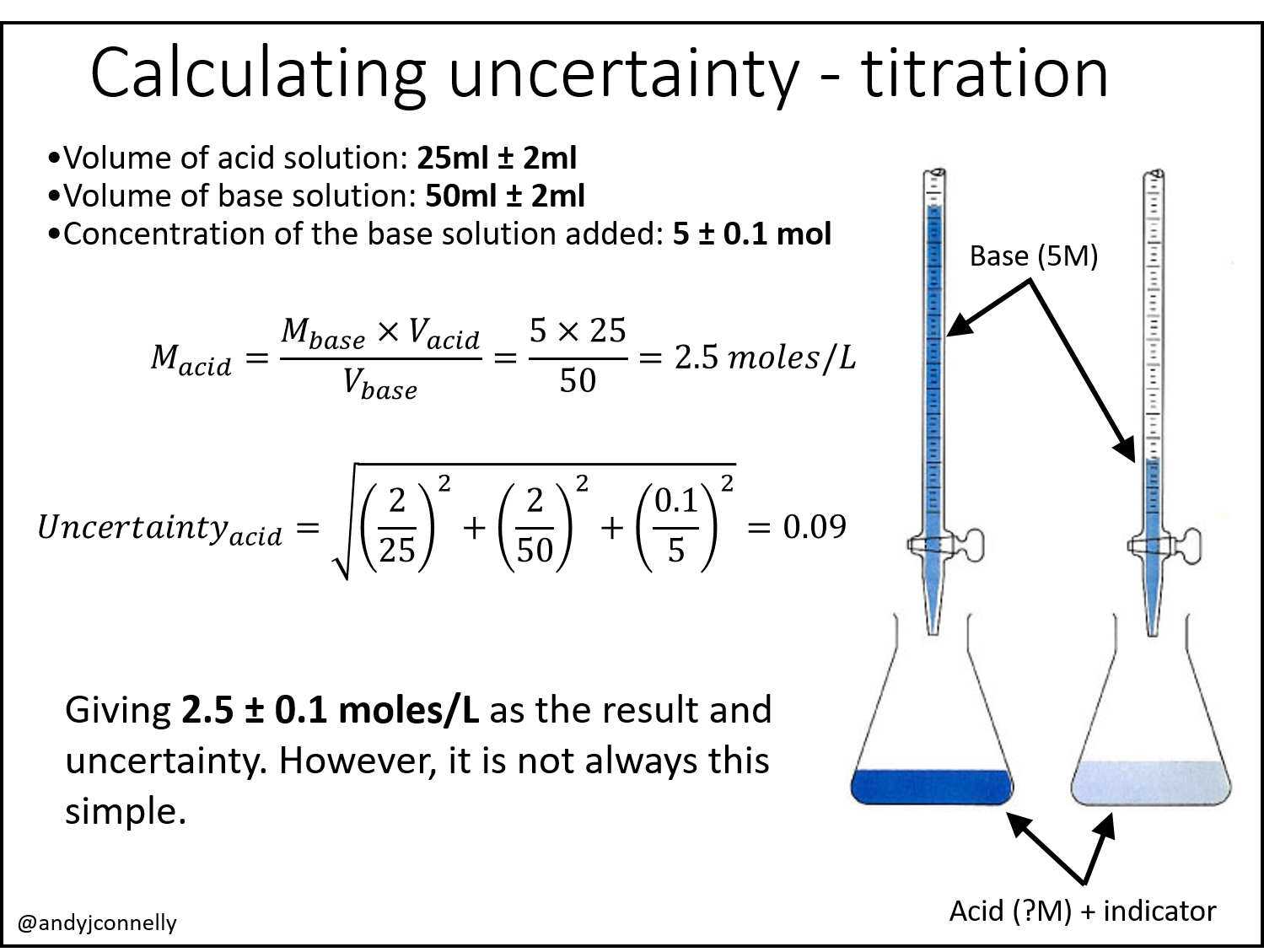
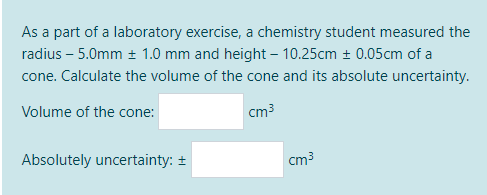The ± 1 second is called the absolute uncertainty Every measurement has an uncertainty or error. e.g. time = 5 seconds ± 1 second There are three main. - ppt download

analytical chemistry - How to deal with exact numbers on error propagation? - Chemistry Stack Exchange
![Analytical Chemistry: Absolute Uncertainty] I am in desperate help to answer this question. I figured out how to solve for Absolute Uncertainty when it is an addition or multiplication. However, I have Analytical Chemistry: Absolute Uncertainty] I am in desperate help to answer this question. I figured out how to solve for Absolute Uncertainty when it is an addition or multiplication. However, I have](https://preview.redd.it/48a3pkftjiq51.png?auto=webp&s=9a03f2bf8b86ce0cd0c14deb155a5d31381d296f)
Analytical Chemistry: Absolute Uncertainty] I am in desperate help to answer this question. I figured out how to solve for Absolute Uncertainty when it is an addition or multiplication. However, I have



![11.1 Determine the uncertainties in results [SL IB Chemistry] - YouTube 11.1 Determine the uncertainties in results [SL IB Chemistry] - YouTube](https://i.ytimg.com/vi/B7dKWE-0FZY/maxresdefault.jpg)







![11.1 State uncertainties as absolute and percentage uncertainties [SL IB Chemistry] - YouTube 11.1 State uncertainties as absolute and percentage uncertainties [SL IB Chemistry] - YouTube](https://i.ytimg.com/vi/wrnCMUP8V1o/maxresdefault.jpg)